Answers
Answer: I think its 32 I'm not sure
Explanation:
Related Questions
calculate the pH of the solution obtained if 40cm^3 of 0.2M HCl was added to 30cm^3 of 0.1M NaOH
Answers
To calculate the pH of the solution obtained by mixing HCl and NaOH, we need to consider the neutralization reaction between the two compounds. The reaction between HCl (hydrochloric acid) and NaOH (sodium hydroxide) produces water (H₂O) and forms a salt (NaCl).
Given:
Volume of HCl solution (V₁) = 40 cm³
Concentration of HCl solution (C₁) = 0.2 M
Volume of NaOH solution (V₂) = 30 cm³
Concentration of NaOH solution (C₂) = 0.1 M
1. Determine the moles of HCl and NaOH used:
Moles of HCl = Concentration (C₁) × Volume (V₁)
Moles of HCl = 0.2 M × 0.04 L (converting cm³ to L)
Moles of HCl = 0.008 mol
Moles of NaOH = Concentration (C₂) × Volume (V₂)
Moles of NaOH = 0.1 M × 0.03 L (converting cm³ to L)
Moles of NaOH = 0.003 mol
2. Determine the limiting reagent:
The stoichiometry of the reaction between HCl and NaOH is 1:1, meaning that they react in a 1:1 ratio. Whichever reactant is present in a smaller amount will be the limiting reagent.
In this case, NaOH is present in a smaller amount (0.003 mol), which means it will be fully consumed during the reaction.
3. Determine the excess reagent and its remaining moles:
Since NaOH is the limiting reagent, we need to find the remaining moles of HCl.
Moles of HCl remaining = Moles of HCl initially - Moles of NaOH reacted
Moles of HCl remaining = 0.008 mol - 0.003 mol
Moles of HCl remaining = 0.005 mol
4. Calculate the concentration of HCl in the resulting solution:
Volume of resulting solution = Volume of HCl solution + Volume of NaOH solution
Volume of resulting solution = 0.04 L + 0.03 L
Volume of resulting solution = 0.07 L
Concentration of HCl in the resulting solution = Moles of HCl remaining / Volume of resulting solution
Concentration of HCl in the resulting solution = 0.005 mol / 0.07 L
Concentration of HCl in the resulting solution ≈ 0.071 M
5. Calculate the pH of the resulting solution:
pH = -log[H⁺]
pH = -log(0.071)
Using logarithm properties, we can determine the pH value:
pH ≈ -log(0.071)
pH ≈ -(-1.147)
pH ≈ 1.147
Therefore, the pH of the solution obtained by mixing 40 cm³ of 0.2 M HCl and 30 cm³ of 0.1 M NaOH is approximately 1.147.
What is the r.a.m. value of krypton (Kr)?A. 36 amuB. 1/36 amuC. 84 amuD. 1/84 amu
Answers
Explanation:
The relative atomic mass is the ratio of the mass of an element compared to the mass of the carbon-12. The atomic mass of krypton is 83.798u.
Answer:
Therefore, the correct answer is A.
Some patterns of electron configuration are listed below. In each case, [G] stands for a noble-gas core and n, m, or o stand for integer numbers like 1, 2, 3 and so on. Think about the chemical elements made of atoms with an electron configuration that matches each pattern. In what block or blocks of the Periodic Table would these elements lie? Would they be metals, nonmetals, metalloids, some of each? Use your answers to these questions to complete the table.a. [G]ns^2np^5b. [G]ns^2np^2c. [G]ns^2mf^14
Answers
Answer:
G]ns^2np^5 group 17 (p-block)
G]ns^2np^2 group 14 (p-block)
G]ns^2mf^14 group 16 (f-block)
Explanation:
The outermost electronic configuration of an element shows the group to which it belongs in the periodic table as shown above in the answer. In addition, to that, we can be able to know from its electronic configuration, whether the element is a metal or not.
For instance;
G]ns^2mf^14 is a rare earth metal, G]ns^2np^2 group 14 is a metalloid while G]ns^2np^5 group 17 is a nonmetal.
What is the specific heat of a substance if a mass of 10.0 kg increases in temperature from 10.0°c to 70.0°c when 2,520 j of heat is applied? use q equals m c subscript p delta t.. 0.00420 j/(gi°c) 0.00661 j/(gi°c) 238 j/(gi°c) 252 j/(gi°c)
Answers
Answer:
0.00420
Explanation:
The equation is Q=mc(T(final)-T(initial), where c is the specific heat, Q is heat supplied, m is mass, T(final) is final temperature and T(initial) is initial temperature (you'll see this written as delta T, which means change in temperature).
2520 = Q
m = 10.0kg; answer choices are in g, not kg, so multiply by 1000 to get m in g; m = 10000 g
Plug in the values you have and solve for c.
2520=(10000)(c)(70-10)
2520=600000c
c=0.0042 j/(gc)
Answer:
4.2 j/kg-C = .0042 j / (g-C)
Explanation:
Specific heat units are j / kg-C
2520 J / [ (10 kg)(70-10 C)] = 4.2 j / kg-C
A sample of carbon dioxide gas (CO2) contains 6 x 1022 molecules. How many moles of carbon dioxide does this represent?
Answers
Answer:
Explanation:
1 ST METHOD
GIVEN DATA:
number of molecules=6.022×10²²molecules
Avogodro's number Na=6.022×10²²
TO FIND:
number of moles
SOLUTION:
As we knom that number of moles=number of molecules/Na
number of moles=6.022×10²²/6.022×10²²
number of moles=1 moles
2ND METHOD:
AS WE KNOW THAT A MOLE OF A SUBSTANCE CONTAINS 6.022×10²² PARTICLES OF THAT SUBATANCE SO
1MOLE OF CO2=6.022×10²²MOLECULES OF CO2
A sample of helium occupies a volume of 160cm3 at 100 KPa and 25°c. what volume will it occupy if the pressure is adjusted to 80 KPa and the temperature remains unchanged?
Answers
Answer:
Explanation:Explore this page
About the gas laws calculator
This is an ideal gas law calculator which incorporates the Boyle's law , Charles's law, Avogadro's law and Gay Lussac's law into one easy to use tool you can use as a:
Boyle's Law-
\(\:\:\:\:\:\:\:\:\:\:\:\star\:\sf \underline{ P_1 \: V_1=P_2 \: V_2}\\\)
(Pressure is inversely proportional to the volume)
Where-
\(\sf V_1\) = Initial volume\(\sf V_2\) = Final volume\(\sf P_1\) = Initial pressure\(\sf P_2\) = Final pressureAs per question, we are given that -
\(\sf V_1\) = 160 cm³\(\sf P_1\) = 100KPa\(\sf P_2\) = 80KPaNow that we have all the required values and we are asked to find out that volume which will be occupied if the pressure is adjusted to 80 KPa and the temperature remains unchanged. For that we can put the values and solve for the final volume of helium-
\(\:\:\:\:\:\:\:\:\:\:\:\:\:\:\:\star\:\sf \underline{ P_1 \: V_1=P_2 \: V_2}\)
\(\:\:\:\: \:\:\:\:\:\:\longrightarrow \sf 100 \times 160 = 80 \times V_2\\\)
\( \:\:\:\:\:\:\:\:\:\:\longrightarrow \sf V_2 = \dfrac{100 \times 160}{80}\\\)
\( \:\:\:\:\:\:\:\:\:\:\longrightarrow \sf V_2 =100\times \cancel{\dfrac{ 160}{80}}\\\)
\(\:\:\:\: \:\:\:\:\:\:\longrightarrow \sf V_2 = 100 \times 2\\\)
\( \:\:\:\:\:\:\:\:\:\:\longrightarrow \sf \underline{V_2 = 200 \:cm^3 }\\\)
Therefore, 200 cm³ will be occupied if the pressure is adjusted to 80 KPa and the temperature remains unchanged.How many grams of a 14.0% (w/w) sugar solution contain 62.5
of sugar?
Answers
Answer:
\(m_{solution}=446.4g\)
Explanation:
Hello,
In this case, by-mass percent is computed in terms of the mass of the solute and the mass of the solvent as shown below:
\(\% m=\frac{m_{solute}}{m_{solution}} *100\%\)
Thus, solving for the mass of the solution, we obtain:
\(\frac{x}{y} m_{solution}=\frac{m_{solute}}{\% m}*100\%\\ \\m_{solution}=\frac{62.5g}{14.0\%}*100\% \\\\m_{solution}=446.4g\)
Regards.
Draw the product you expect from the reaction of (R)-2-bromooctane with -CH3CO2- and Br-.. Use the wedge/hash bond tools to indicate stereochemistry where it exists. Include H atoms at chiral centers only. If a group is achiral, do not use wedged or hashed bonds on it.
Answers
From the mechanism shown, the major product of the reaction is (S)-(+)-2-octanol.
What is an SN2 reaction?An SN2 reaction is one in which there is an inversion of configuration and the attack of the nucleophile occurs from behind the substrate.
As shown in the mechanism attached here, the major product of this reaction is (S)-(+)-2-octanol.
Learn more about SN2 reaction:https://brainly.com/question/14080839
#SPJ1

a. Identify the structures shown in the diagram. b. Identify the information that is contained within these structures. c. Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person. d. Explain why the structures are in pairs.
Answers
The answer responses to the structures shown in the diagram are:
A. chromosomes
C. They would be the same.
B. They are in pairs because each one comes from a different parent.
What is the structure about?The chromosomes are in pairs because humans have a diploid number of chromosomes, meaning they have two sets of chromosomes, one inherited from each parent.
The nucleus is important in eukaryotic cells and has many important parts that help the cell work properly. There are some parts inside cells called the nuclear membrane, nucleoplasm, nucleolus, and chromatin. Chromatin is made up of DNA and other proteins.
Every part of a person's body has the same genes, but the way they are organized can be different in different types of cells. The chromosomes in our skin cells might not be the same as the chromosomes in our muscle cells, even if they come from the same person.
Learn more about nucleus from
https://brainly.com/question/9376695
#SPJ1
Identify the structures shown.
A. chromosomes
B. mitochondria
C. nuclei
D. vacuoles
C
Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person.
A. There would be longer.
B. They would be shorter.
C. They would be the same.
D. They would be different.
Describe how the structures from this cell would compare to the structures in the nucleus of another body cell from the same person.
A. There would be longer.
B. They would be shorter.
C. They would be the same.
D. They would be different.
Explain why the structures are in pairs.
A. They aren't in pairs.
B. They are in pairs because each one comes from a different parent.
C. This cell is making a copy of itself.
D. The cell always has 2 copies in case 1 is damaged.
How does the chemical structure of a substance affect its interaction with other substances?
Answers
This is due to the fact that a substance's chemical qualities, such as its molecular form, polarity, and functional groups, govern how it behaves and interacts with other substances.
How does their chemical makeup impact their chemical characteristics?By illustrating the spatial arrangement of atoms and chemical bonds within the molecule, chemical structure establishes the molecular geometry of a compound. In doing so, chemists are given a crucial visual depiction of a chemical formula.
In what ways do drugs interact with one another?In a chemical reaction, reactants come into contact with one another, atoms in the reactants break their connections with one another, and then the atoms reorganise and form new bonds to create the products.
To know more about molecule visit:-
https://brainly.com/question/19556990
#SPJ1
what is the change in mass of A in
60 minutes?
Mass of A (g)
12.4
10.4
9.1
7.7
6.2
Time
O
15
30
45
60
Answers
Answer:
To determine the change in mass of A over the given time period, we need to find the difference between the initial mass of A and the final mass of A.
From the given table, we can see that the initial mass of A at t = 0 (start time) is 12.4 g and the final mass of A at t = 60 minutes (end time) is 6.2 g.
Therefore, the change in mass of A over 60 minutes is:
Final mass of A - Initial mass of A
= 6.2 g - 12.4 g
= -6.2 g
The negative sign indicates that the mass of A decreased over time, which means that A underwent some kind of reaction or process that caused it to lose mass.
The change in mass of A over 60 minutes is -6.2 grams.
To determine the change in mass of A over 60 minutes, we need to compare the initial mass to the final mass.
From the given information, we can see that the mass of A decreases over time.
Let's calculate the change in mass.
Initial Mass of A: 12.4 g
Final Mass of A: 6.2 g
Change in Mass of A = Final Mass of A - Initial Mass of A
= 6.2 g - 12.4 g
= -6.2 g
The change in mass of A over 60 minutes is -6.2 grams.
Note that the negative sign indicates a decrease in mass.
For such more questions on mass
https://brainly.com/question/1838164
#SPJ8
If 2.5 ggiven off? of KCLO3 were heated in a test tube, how many grams of oxygen gas should be
Answers
Answer:
1.46878825 g O_3
Explanation:
the balanced equation is:
2 KClO_3 = 2 KCl + 3 O_2
using stoichiometry, you can find the amount of O_2 produced from 2.5 g KClO_3
2.5 g KClO_3 x (1 mol / 122.55 g KClO_3) x (3 mol O_3 / 2 mol KClO_3) x (48 g O_3 / 1 mol O_3) = 1.46878825 g O_3
11 . A guest orders a drink that contains 4 1/2 ounces of 80-proof liquor. Approximately how many drinks does this beverage contain?
Answers
From the calculations that have been done, there are 3 drinks.
How many drinks are there?The information that we have been provided here is very important. We can obtain the amount of alcohol by looking at the fact that it contains 80-proof liquor.
This 80-proof liquor. is an equivalent of 40% alcohol this means that there is 1.5 ounce of distilled spirit.
Thus, the number of drinks there is;
4.5/1.5 = 3
Learn more about alcohol:https://brainly.com/question/24255571
#SPJ1
Which functional group is within the compound shown below?

Answers
Most acids are strong acids.
A. True
B. False
Answers
Answer:
False, There are only a few strong acids, so many people choose to memorize them. All the other acids are weak
Explanation:
Net ionic equation for potassium sulfide and magnesium iodide
Answers
The net ionic equation for the reaction between potassium sulfide and magnesium iodide is S2- + Mg2+ -> MgS, as the potassium and iodide ions are spectator ions and do not participate in the reaction.
To determine the net ionic equation for the reaction between potassium sulfide (K2S) and magnesium iodide (MgI2), we first need to identify the ions present in each compound and then determine the products formed when they react.
Potassium sulfide (K2S) dissociates into two potassium ions (K+) and one sulfide ion (S2-):
K2S -> 2K+ + S2-
Magnesium iodide (MgI2) dissociates into one magnesium ion (Mg2+) and two iodide ions (I-):
MgI2 -> Mg2+ + 2I-
Now, we need to determine the possible products when these ions combine. Since potassium (K+) has a +1 charge and iodide (I-) has a -1 charge, they can combine to form potassium iodide (KI):
K+ + I- -> KI
Similarly, magnesium (Mg2+) and sulfide (S2-) can combine to form magnesium sulfide (MgS):
Mg2+ + S2- -> MgS
Now, we can write the complete ionic equation by representing all the ions present before and after the reaction:
2K+ + S2- + Mg2+ + 2I- -> 2KI + MgS
To obtain the net ionic equation, we remove the spectator ions, which are the ions that appear on both sides of the equation and do not participate in the actual reaction. In this case, the spectator ions are the potassium ions (K+) and the iodide ions (I-).
Thus, the net ionic equation for the reaction between potassium sulfide and magnesium iodide is:
S2- + Mg2+ -> MgS
For more such questions on ionic equation visit:
https://brainly.com/question/25604204
#SPJ8
7) How many molecules of CO2 are in 2.5 L at STP?
Answers
By using the ideal gas law and Avogadro's number, we find that there are approximately 6.72 × 10^22 molecules of CO2 in 2.5 L at STP.
To determine the number of molecules of CO2 in 2.5 L at STP (Standard Temperature and Pressure), we can use the ideal gas law and Avogadro's number.
Avogadro's number (N_A) is a fundamental constant representing the number of particles (atoms, molecules, ions) in one mole of substance. Its value is approximately 6.022 × 10^23 particles/mol.
STP conditions are defined as a temperature of 273.15 K (0 °C) and a pressure of 1 atmosphere (1 atm).
First, we need to convert the volume from liters to moles of CO2. To do this, we use the ideal gas law equation:
PV = nRT,
where P is the pressure, V is the volume, n is the number of moles, R is the ideal gas constant, and T is the temperature in Kelvin.
Since we have STP conditions, we can substitute the values:
(1 atm) × (2.5 L) = n × (0.0821 L·atm/(mol·K)) × (273.15 K).
Simplifying the equation:
2.5 = n × 22.4149.
Solving for n (the number of moles):
n = 2.5 / 22.4149 ≈ 0.1116 moles.
Next, we can calculate the number of molecules using Avogadro's number:
Number of molecules = n × N_A.
Number of molecules = 0.1116 moles × (6.022 × 10^23 particles/mol).
Number of molecules ≈ 6.72 × 10^22 molecules.
Therefore, there are approximately 6.72 × 10^22 molecules of CO2 in 2.5 L at STP.
For more such questions on ideal gas law visit:
https://brainly.com/question/27870704
#SPJ8
If mercury barometer is replaced by water barometer, height of water column
i. will be less than that of Hg Column
ii. will be greater than that of Hg column iii. will be equal to that of Hg column
iv. will be none of these
Answers
Answer:
answer is first one 1 will be less then that of hg coloumn
How many electrons, protons and neutrons does the element Argon have?
Answers
Answer:
18 Protons
18 Neutrons
&
18 Electrons
The heat of vaporization of water is 540 cal/g. How much heat is needed to change 4.28g of water to steam?
Answers
Answer:
2311.2 cal
Explanation:
540 cal / g * 4.28 g = 2311.2 cal
2311.2 cal heat is needed to change 4.28g of water to steam.
What is heat?Heat is the transfer of kinetic energy from one medium or object to another, or from an energy source to a medium or object.
The heat of vaporization is defined as the amount of heat needed to turn 1g of a liquid into a vapour, without a rise in the temperature of the liquid.
The heat of vaporization is equal to the thermal energy required for vaporization divided by the mass of the substance that is vaporizing. Its formula is Hv = q/m.
Putting the values in the formula:
540 cal / g x 4.28 g = 2311.2 cal
Hence, 2311.2 cal heat is needed to change 4.28g of water to steam.
Learn more about the heat here:
https://brainly.com/question/13411214
#SPJ2
Find the mass, in grams, of 4.10x1023 molecules of F2.
Answers
Answer:
25.88 g of F₂.
Explanation:
From the question given above, the following data were obtained:
Number of molecules of F₂ = 4.10×10²³ molecules
Mass of F₂ =?
From Avogadro's hypothesis,
1 mole of F₂ = 6.02×10²³ molecules
Next, we shall determine the mass of 1 mole of F₂. This can be obtained as follow:
1 mole of F₂ = 2 × 19 = 38 g
Thus,
38 g of F₂ = 6.02×10²³ molecules
Finally, we shall determine the mass of 4.10×10²³ molecules of F₂. This can be obtained as follow:
6.02×10²³ molecules = 38 g of F₂
Therefore,
4.10×10²³ molecules = 4.10×10²³ × 38 / 6.02×10²³
4.10×10²³ molecules = 25.88 g
Thus, 25.88 g of F₂ contains 4.10×10²³ molecules.
prop-1-yne + 2HBr/H2O2 = A;
A + 2H2O = B;
B + K2CO3(aq) = C;
C + heat = D;
D + HBr = E.
find the compounds A, B, C, D and E
Answers
Based on the given reactions, the compounds are as follows:
A: The specific product formed from the reaction between prop-1-yne and either 2HBr or H2O2.
B: The product formed when compound A reacts with 2H2O.
C: The product formed when compound B reacts with K2CO3(aq).
D: The product formed from the heat-induced reaction of compound C.
E: The product formed when compound D reacts with HBr.
Based on the given reactions, let's analyze the compounds involved:
Reaction 1: prop-1-yne + 2HBr/H2O2 = A
The reactant prop-1-yne reacts with either 2HBr or H2O2 to form compound A. The specific product formed will depend on the reaction conditions.
Reaction 2: A + 2H2O = B
Compound A reacts with 2H2O (water) to form compound B.
Reaction 3: B + K2CO3(aq) = C
Compound B reacts with K2CO3(aq) (potassium carbonate dissolved in water) to form compound C.
Reaction 4: C + heat = D
Compound C undergoes a heat-induced reaction to form compound D.
Reaction 5: D + HBr = E
Compound D reacts with HBr (hydrobromic acid) to form compound E.
For more such questions on compounds
https://brainly.com/question/704297
#SPJ8
b. How many kJ of heat are needed to completely vaporize 50.0g of water at 100°C? [Ans:113. kJ]
Answers
The amount, in kJ, of heat needed to completely vaporize 50.0g of water at 100°C is 118.8 kJ.
Heat of vaporization of waterThe heat needed to completely vaporize 50.0g of water at 100°C can be calculated using the following formula:
q = m x Hv
where:
q is the heat needed in joules (J)m is the mass of water in grams (g)Hv is the heat of vaporization of water which is approximately 40.65 kJ/mol at standard temperature and pressure.First, we need to convert 50.0g to moles by dividing by the molar mass of water which is approximately 18.015 g/mol3:
moles of water = 50.0 g / 18.015 g/mol moles of water = 2.776 mol
Thus:
q = (2.776 mol) x (40.65 kJ/mol) q = 112.8 kJ
In other words, 112.8 kJ of heat is needed to completely vaporize 50.0g of water at 100°C.
More on heat of vaporization can be found here: https://brainly.com/question/12625048
#SPJ1
Suppose that you place an object along the principal axis of a concave spherical mirror, exactly halfway to the center of the curvature (half of the radius). What will the result be?
Answers
Answer:
There will be no clear image of the object.
Explanation:
For a concave spherical mirror, we have the distance from center of curvature to the mirror is the radius of curvature, R, and half of the length of the radius of curvature is the focal length, f, of the mirror, that is we have;
\(f = \dfrac{R}{2}\)
From the equation of a mirror in optics we have;
\(\dfrac{1}{f} = \dfrac{1}{d_o} + \dfrac{1}{d_i}\)
Where:
\(d_o\) = Distance of the object from the mirror
\(d_i\) = Distance of the image from the mirror
Hence, where the object distance is half the radius of curvature or f, we have;
\(\dfrac{1}{f} = \dfrac{1}{f} + \dfrac{1}{d_i}\)
Therefore, the location of the image formed will be at;
\(\dfrac{1}{d_i}= \dfrac{1}{f} - \dfrac{1}{f} = 0\)
\(d_i = \dfrac{1}{0}= \infty\)
Hence, since the location of the image formed will be at infinity there will be no clear image of the object.
Answer:
A clear image of that object won't be formed at all.
Explanation:
PF The point exactly halfway to the center of curvature of a concave spherical mirror is called the center of curvature. The point exactly halfway from the mirror to the center is called the focal point (or focus) of the mirror. When the object is exactly at the focus of the mirror, the reflecting rays are parallel and therefore won't meet to form an image.
Hard water often contains dissolved Ca^2+ and Mg^2+ ions. One way to soften water is to add phosphates. The phosphate ion forms insoluble precipitates with calcium and magnesium ions, removing them from solution. A solution is 0.055 MM in calcium chloride and 0.075 MM in magnesium nitrate. What mass of sodium phosphate would you add to 1.0 LL of this solution to completely eliminate the hard water ions? Assume complete reaction.
Answers
Answer:
14.2g of sodium phosphate are required
Explanation:
To solve this question we need to find the moles of Ca²⁺ and Mg²⁺ ions. And based on the reactions:
3Ca²⁺ + 2PO₄³⁻ → Ca₃(PO₄)₂ (s)
3Mg²⁺ + 2PO₄³⁻ → Mg₃(PO₄)₂ (s)
we can find the moles of phosphate required to precipitate all these ions and its mass:
Moles Ca²⁺:
1.0L * (0.055mol / L) = 0.055mol
Moles Mg²⁺:
1.0L * (0.075mol / L) = 0.075mol
Total moles = 0.13 moles of ions
Moles of phosphate ion required:
0.13 moles * (2 moles PO₄³⁻ / 3 moles ions) = 0.0867 moles PO₄³⁻
The moles of sodium phosphate (Na₃PO₄) are = 0.0867 moles
The mass is -Molar mass Na₃PO₄: 164g/mol-:
0.0867 moles Na₃PO₄ * (164g / mol) =
14.2g of sodium phosphate are requiredGive the IUPAC name for the following molecule.
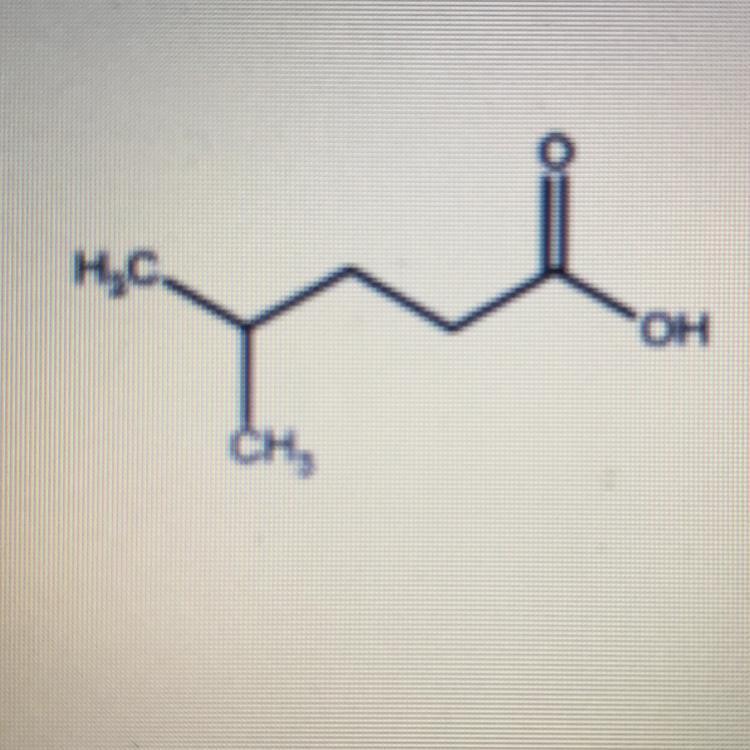
Answers
Answer:
The IUPAC name is:
4-methyl pentanoic acid
Explanation:
4-Methylpentanoic acid is actually a carboxylic acid. It has five carbons and there is a methyl substitution which is at the fourth carbon.
Molecular Formula: \(C_{6}H_{12}O_{2}\)
The acid is known to be a metabolite of 20 alpha-hydroxyl cholesterol. From the excerpt, the acid is a branched-chain and also known as a saturated fatty acid. It is known to be a conjugate acid of an isocaproate.
Other names for the acid are also called 4-methylvaleric acid or isocaproic acid.
What does the term "basic unit of matter" refer to?
O A.
Atoms
ОВ.
Elements
O c. Molecules
Answers
Hope your day is going well
Atoms are referred to basic unit of matter
Hope this helps *smiles*
Explanation:
The term "basic unit of matter "refers to atom
A Atom
plz help ❤️
Explain how the pH of water has a synergistic relationship with other substances.
Provide an example.
Answers
Answer:
Explanation:
he pH of the water will determine the toxic effects, if any, of these substances.
Pure water is neutral with the pH of 7. But when water combines with other substance the pH varies as per the acidity of the combining substance.
What is pH?pH is the measure of acidity of a solution and mathematically it is the negative logarithm of hydrogen ion concentration. Acidic substance have a pH of less than 7 whereas, basic substance have higher pH value.
Water when combines with some acids, its acidity increases. For example acid rain resulting from the reaction of rain water with oxides of nitrogen and Sulphur forming nitric acid and sulphuric acid.
pH of water have a role in the toxic effects of components in water pH and alkalinity of water determines the coagulation of other substances in water.
Hence, the properties of other substance in water is pH dependent and might effect the alkalinity or acidity of water as well the properties of substances in water such as toxicity.
To learn more about pH , refer the link below:
https://brainly.com/question/2288405
#SPJ2
Write the net ionic equation for the precipitation of calcium sulfide from aqueous solution
Answers
Answer:
Answer is Ca2+(aq)+S2-(aq)=>CaS(s)
Explanation:
I hope it's helpful!
The net ionic equation for the precipitation of calcium sulfide in solution is; Ca^2+(aq) + S^2-(aq) -------> CaS(s)
Let me give a typical instance since your question is incomplete. Imagine an aqueous solution of H2S and CaCl2 from which calcium sulfide does precipitate.
The molecular reaction equation is;
H2S(aq) + CaCl2(aq) -------> CaS(s) + 2HCl(aq)
The complete ionic equation is;
2H^+(aq) + S^2-(aq) + Ca^2+(aq) + 2Cl^-(aq) -------> CaS(s) + 2H^+(aq) + 2Cl^-(aq)
The net ionic equation is;
Ca^2+(aq) + S^2-(aq) -------> CaS(s)
Learn more: https://brainly.com/question/22885959
Find the number of grams in 3.45 x 10^21 molecules NiO
Answers
Answer:
Mass = 0.43 g
Explanation:
Given data:
Mass in gram = ?
Number of molecules of NiO = 3.45×10²¹
Solution:
The given problem will solve by using Avogadro number.
It is the number of atoms , ions and molecules in one gram atom of element, one gram molecules of compound and one gram ions of a substance.
The number 6.022 × 10²³ is called Avogadro number.
1 mole = 6.022 × 10²³ molecules
3.45×10²¹ molecules × 1 mol / 6.022 × 10²³ molecules
0.57×10⁻² mol
0.0057 mol
Mass in gram:
Mass = number of moles × molar mass
Mass = 0.0057 mol × 74.69 g/mol
Mass = 0.43 g