why is it more efficient in a liquid liquid extraction to do multiple extractions rather than one large one
Answers
In liquid-liquid extraction, it is more efficient to do multiple extractions rather than one large one because the solubility of the solute in the solvent may decrease in each extraction.
The amount of solute that dissolves in a solvent decreases with each extraction. Multiple extractions are performed to extract the maximum amount of solute from the mixture being separated in liquid-liquid extraction.
What is liquid-liquid extraction?Liquid-liquid extraction is a technique that is used to isolate one or more dissolved or suspended components from a mixture based on their relative solubilities in two immiscible liquids.
What is multiple extractions?Multiple extractions, also known as re-extraction, is a procedure that involves separating a target compound from a mixture by extracting it several times with the same solvent or a series of solvents.
Multiple extractions are done when the solubility of the solute in the solvent decreases with each extraction. This will help to extract the maximum amount of solute from the mixture.
To know more about multiple extractions click on below link :
https://brainly.com/question/31322526#
#SPJ11
Related Questions
True or False a. Sodium chloride is the main salt found in sea water. ……………………………………………………………………. b. Potassium bromide salt is not found in sea water ……………………………………………………………………. c. Both evaporation and electric conduction techniques prove that sea water contain the dissolved salts. …………………………………………………………………….. d. The amount of fresh water is greater than 5% of the total Earth water. …………………………………………………………………….. e. In water treatment process, filter membrane is used to remove fine particles and other impurities from water. …………………………………………………………………………....
Answers
Answer:
A is true NaCl
B is false K2Br
C is true
D false its only 0.03 percent
E is true
How many moles of 02 are produced by the decomposition of 6.2 moles of KCLO3 Balance reaction first _KCLO3 _KCL +_ O2
Answers
Number of moles of O2 produced by the decomposition of 6.2 moles of KCLO3 is 9.3 moles.
The mole idea is a useful way to indicate how much of a substance there is. Any measurement can be divided into two components: the magnitude in numbers and the units in which the magnitude is expressed.
Initial reaction,
KCLO3 ⇒ KCL + O2
The ratio is 1: 1 for KClO3 and O2
After balancing the equation we get,
2KClO3 ⇒ 2KCl + 3O2
So ratio of KClO3 and O2 is now 2:3
For every 2 moles of KClO3 we have 3 moles of O2 so
For 6.2 moles of KCLO3,
Number of moles of O2 = 6.2x3/2
= 9.3 moles
Therefore, number of moles of O2 produced by the decomposition of 6.2 moles of KCLO3 is 9.3 moles.
Learn more about Moles;
https://brainly.com/question/28037816
#SPJ4
PLZ HELP ME WITH MY WORK

Answers
Answer:
c. H2 andO2
I think this is the answer not sure ok
Oh my god sadddddddddddddddddd
Answers
Answer:
sad? that your giving out free points?
Explanation:
Answer:
what wrong
tell i can help u
Explanation:
I NEED THIS ASAP!! Select all that apply.
Air pollution particles may be removed by _____.
oceans
natural cycles
soil
rain
snow
wind
Answers
Answer:
Natural Cycles
Explanation:
Most sulfur oxides are produced by burning: coal
Air pollution particles may be removed by _____. rain snow wind natural cycles
The three primary sources of air pollution are _____. factories cars electric power plants
The two basic types of air pollutants are _____. gases particulates
May I get brainliest please?
If I'm wrong sorry.
Atudent prepared a 0. 10M acetic acid olution and experimentally meaured the pH to be 2. 88. Calculate Ka for acetic acid and determine it percentage diociation?
Answers
The Ka for acetic acid is 1.76 * 10^{-5} and its percentage dissociation is 1.32 %.
What is pH?
pH is a measure of the acidity or alkalinity of a solution. It is measured on a scale from 0 to 14, with 0 being the most acidic, 7 being neutral, and 14 being the most alkaline. A pH of 7 is considered neutral, meaning that the solution is neither acidic nor alkaline.
(i) pH= 2.88
\([H_{3} 0^{+}] = 10^{-2.88} \\ = 1.32 * 10^{-3}\) \(here, {H_{3} }0^{+} = 10^{-pH}\)
\(CH_{3} CO_{2} H + H_{2} O_{2} = CH_{3} CO_{2} ^{-} + H_{3} O^{+}\)
I 0.10 0 0
C -x +x +x
E 0.10-x x x
\(Ka = \frac{[ CH_{3} CO_{2}^{-} ] [ H_{3} O^{+} ]}{ [ CH_{3} CO_{2} H ] }\)
\(= \frac{x^{2} }{0.10-x} \\\\= \frac{[1.32 * 10^{-3}]^{2} }{0.10- [1.32 * 10^{-3}] } \\\\= 1.76 * 10^{-5}\)
(ii) % Ion = \(= \frac{x}{0.10} * 100%\\= \frac{1.32 * 10^{-3} }{0.10} * 100\\\\\)
= 1.32 %
Therefore, 1.76 * 10^{-5} is the Ka for acetic acid and its percentage dissociation is 1.32 %.
To learn more about pH from the link
https://brainly.com/question/15289741
#SPJ4
If excess ammonium sulfate reacts with 20.0g of calcium hydroxide how many liters of ammonia are produced at STP?
(NH4) 2SO4 + Ca (OH) 2 --> CaSO4 + NH3 + H20
Answers
If excess ammonium sulfate reacts with 20.0g of calcium hydroxide then liters of ammonia are produced at STP 0.081L
Ammonium sulphate is the organic solvent and the primary use of ammonium sulphate is as a fertilizer for alkaline soils
Here given reaction is
(NH₄)₂SO₄ + Ca(OH)₂ → CaSO₄ + NH₃ + H₂O
Here given data is
Calcium hydroxide = 20.0g
We have to find liters of ammonia are produced at STP =?
20.0g of (NH₄)₂SO₄ STP = ?
Then 20.0g of (NH₄)₂SO₄×1 mol/132g×0.54L/1 mol = 0.081L
Know more about litre
https://brainly.com/question/2981088
#SPJ1
what is atomic packing of the (100) plane for the fcc crystal structure?
Answers
There are one atom in the center and four atoms at each of the four corners of the (100) plane.
The unit cell contains one-fourth of each corner atom and the complete middle atom, making the total number of atoms on the (100) plane inside the cell N100 = 4 (1/4) + 1 1 = 2.
For more questions like fcc crystal structure visit the link below
https://brainly.com/question/11981666
#SPJ4
17. what intermolecular forces that would be exerted in a 1.0L container of this substance at STP.
a. NH3
b. CHCl3 (Carbon monohydride trichloride - "Chloroform")
Answers
a. NH3 (Ammonia): In a 1.0L container of NH3 at Standard Temperature and Pressure (STP), intermolecular forces such as hydrogen bonding would be exerted.
b) 1.0L container of CHCl3 at STP, dipole-dipole interactions (along with London dispersion forces) would be present.
a. NH3 (Ammonia):
In a 1.0L container of NH3 at Standard Temperature and Pressure (STP), intermolecular forces such as hydrogen bonding would be exerted. Ammonia (NH3) molecules consist of one nitrogen atom bonded to three hydrogen atoms, forming a trigonal pyramidal shape.
Hydrogen bonding is a type of intermolecular force that occurs when a hydrogen atom is covalently bonded to an electronegative atom (in this case, nitrogen) and interacts with another electronegative atom (in this case, the nitrogen atom of a neighboring NH3 molecule).
b. CHCl3 (Chloroform):
In a 1.0L container of CHCl3 (chloroform) at STP, intermolecular forces such as dipole-dipole interactions would be exerted. Chloroform (CHCl3) molecules consist of one carbon atom bonded to three hydrogen atoms and one chlorine atom, forming a tetrahedral shape.
Dipole-dipole interactions arise due to the difference in electronegativity between chlorine and hydrogen atoms. The chlorine atom is more electronegative than hydrogen, resulting in a polar covalent bond.
While dipole-dipole interactions are present in CHCl3, it is worth noting that CHCl3 also exhibits London dispersion forces. These forces occur due to temporary fluctuations in electron distribution, creating temporary dipoles.
London dispersion forces are the weakest type of intermolecular force but still contribute to the overall intermolecular interactions in CHCl3.
For more such questions on Standard Temperature and Pressure visit:
https://brainly.com/question/14820864
#SPJ8
Cabbage juice indicator solutions can show pH values from acidic to basic.
Which color represents the most basic solution?
Which color represents the most acidic solution?
Which color represents a neutral solution?
Answers
More acid solution has red colour, more basic solution has purple colour whereas neutral solution has green colour.
Which color represents the most acidic solution?As the solution becomes more acidic, the color of the solution changes from green toward red.
Which color represents the most basic solution?As the solution becomes more basic, the color changes of the solution from green toward purple.
Which color represents a neutral solution?Green color of the solution represents a neutral solution having pH 7.
So we can conclude that More acid solution has red colour, more basic solution has purple colour whereas neutral solution has green colour.
Learn more about acid here: https://brainly.com/question/25148363
#SPJ1
What is the volume of a container that contains 24. 0 grams of N2 gas at 328K and. 884 atm? Also asks to identify the P, V, N, R, and T. If unknown then put "?"
Answers
The volume of the container that that contains 24. 0 grams of N2 gas at 328K at 884 atm is found to be 2.65 Liters.
The ideal gas equation is given as, PV = nRT, where, P is pressure V is volume, n is the moles, R is the gas constant and T is the temperature.
It is given to us that 24 grams of N₂ is to be stored in the container with 884 atm pressure and 328K temperature.
Now, putting all the values in the equation to find the volume of the container,
884V = 24/28(8.34)(328)
V = 2.65 Liters,
So, the volume of the container will be 2.65 Liters.
To know more about the ideal gas equation, visit,
https://brainly.com/question/27870704
#SPJ4
I onlyI and II onlyII onlyII and III onlyI and III onlyIII only

Answers
The species that act as acid are the ones that donates H⁺ in the reaction.
Since we are working with an equilibrium, we have to consider both directions.
From left to right, we have H₂O turning into OH⁻ because it lost one H⁺, so H₂O is acting as an acid in the forward reaction.
From right to left, we have CH₃NH₃⁺ tunrning into CH₃NH₂ because it lost one H⁺, so CH₃NH₃⁺ is acting as an acid in the backwards reaction.
So, the species that act as acids are H₂O and CH₃NH₃⁺, I and II only.
What happens in an oxidation half-reaction?
ОА. .
Elements gain electrons.
OB.
Elements lose electrons.
Ос. .
Oxygen is added.
OD. Oxygen is lost.
Answers
Answer:
Elements lose electrons.
Explanation:
Which of the statements is a simplified equation for cellular respiration? a. glucose + oxygen - carbon dioxide + water b. lipid + water -> fatty acids + carbon dioxide c. carbon dioxide + water + light -> glucose + oxygen d. glucose + nitrogen -> carbon monoxide + water
Answers
The correct simplified equation for cellular respiration is glucose + oxygen → carbon dioxide + water . i.e. option A
Cellular respiration is defined as the process by which cells convert glucose and oxygen into energy in the form of ATP (adenosine triphosphate). It is a metabolic process that occurs in the mitochondria of eukaryotic cells and in the cytoplasm of prokaryotic cells.
Cellular respiration can be divided into three main stages viz. glycolysis, kreb's cycle and electron transport chain. These are discussed below:
Glycolysis: The first stage of cellular respiration is glycolysis, which takes place in the cytoplasm. In glycolysis, glucose is broken down into two molecules of pyruvate, and a small amount of ATP is produced.
Kreb's cycle: The second stage of cellular respiration is the Krebs cycle, which takes place in the mitochondria. In the Krebs cycle, pyruvate is broken down into carbon dioxide, and more ATP is produced.
Electron transport chain: The final stage of cellular respiration is the electron transport chain, which also takes place in the mitochondria. In the electron transport chain, electrons from the Krebs cycle are used to produce a large amount of ATP.
Thus, it is estimated that cellular respiration produces a net gain of 36-38 ATP molecules per molecule of glucose. The process of cellular respiration is essential for providing cells with the energy they need to carry out their functions and is an important part of the metabolism of all living organisms.
Therefore, the statement that represents a simplified equation for cellular respiration is option A.
To know more about cellular respiration here
https://brainly.com/question/30791350
#SPJ4
The following information is known about a loan. Time = 4 years Interest rate = 3. 1% Original amount of loan = $1,200 What amount of simple interest will be paid on the loan? $37. 20 $148. 80 $1. 51. 20 $1,848,80
Answers
If the time is 4 years interest rate is 3.1% and the original amount of loan is $1,200 then the simple interest that will be paid on the loan is $148.80.
Simple interest, also represented as SI, can be defined as the amount of interest for a particular amount of money at a certain rate of interest.
Principal = 1200
Rate = 3.1% (.031)
Time = 4 years
To calculate the simple interest,
Simple interest (I) = principle x rate x time
Where, principal is the initial principal balance.
Rate is the annual interest rate and time is the duration of money borrowed.
= 1200 x .031 x 4
= $148.80
To learn more about simple interest here
https://brainly.com/question/25845758
#SPJ4
after studying the causes of physical weathering in the table on the next page, infer wich type of weathering caused the rock shown above to split.explain your reasoning
Answers
Physical weathering is caused by the effects of temperature changes on rocks, causing them to break apart. The process is sometimes aided by water.
Abrasion Weather effects such as wind and water break rocks into small pieces. Exfoliated weathered rock expands and fractures when pressure is released from unloading. Frostwedge rock cracks when water freezes and expands. Chemical weathering involves the interaction of rocks with mineral solutions (chemicals), changing the composition of rocks.
Rocks are formed by weathering and erosion that break down existing rocks through physical and chemical processes. Examples of chemical weathering include rust caused by oxidation and acid rain caused by carbonic acid dissolving rocks. Other Chemical Weathering B. Dissolution. Rocks and minerals break down to form soil.
Learn more about Weathering here:-https://brainly.com/question/829782
#SPJ9
small cations are attracted to colloid surfaces more strongly than large cations.
Answers
Colloids are particles that measure between 1 and 1000 nanometers in size and scatter light. They are relatively stable particles that remain suspended in a solvent and do not settle over time.
Because of their small size, colloids have a large surface area, which is a key factor in their reactivity with ions, including cations.The attraction of ions towards a colloid surface is based on their size, charge, and concentration. The charge of a cation plays a vital role in the interaction of a colloid's surface. This is because a colloid's surface carries an opposite charge to that of the cation.
Small cations have a higher charge density than larger ones, which means they are attracted more strongly to colloid surfaces.The high charge density of small cations can interact with the colloid's surface at a closer range than large cations. Because of their size, large cations cannot come as close to the colloid's surface as small cations. Hence, small cations are attracted to colloid surfaces more strongly than large cations.As a result, smaller cations are more attracted to the surface of the colloids than larger ones. This suggests that the stability of colloidal systems is affected by the size of the cation involved.
To know more about Colloids visit;-
https://brainly.com/question/30560048
#SPJ11
what is relative abundance isotopes
Answers
The relative abundance of isotopes is the number of atoms of a particular isotope divide by the total number of atoms of all isotopes of that element, multiplied 100 percent.
What is relative abundance isotopes?The relative abundance of an isotope is the percentage of atoms with a specific atomic mass found in a naturally occurring sample of an element.
Also relative abundances refers to the relative proportions of the stable isotopes of each element. They are most often quoted as atom percentages
To calculate the percent abundance of each isotope in a sample of an element, the number of atoms of a particular isotope is usually divide by the total number of atoms of all isotopes of that element and then multiply the result by 100 since it is expressed in percentage.
Mathematically, the formula for relative abundance is given as;
R.A = ( number of atoms of isotope / total number of atoms ) x 100%
Learn more about relative abundance here: https://brainly.com/question/6844925
#SPJ1
what volume of oxygen gas reacts with 20.0 ml of hydrogen chloride?
Answers
The volume of oxygen gas that reacts with 20.0 ml of hydrogen chloride is 3.0 ml. The reaction of hydrogen chloride (HCl) and oxygen (O2) can be represented as follows: 4HCl + O2 → 2H2O + 2Cl2
To answer this question, we will use the balanced chemical equation and the ideal gas law. The volume of a gas is directly proportional to the number of moles of that gas at a constant temperature and pressure. The ideal gas law can be represented as PV = nRT, where P is the pressure, V is the volume, n is the number of moles, R is the ideal gas constant, and T is the temperature. We can rearrange this equation to solve for the number of moles of a gas, which is n = PV/RT.
We know the volume of hydrogen chloride and the balanced chemical equation, so we can calculate the number of moles of hydrogen chloride. Then, we can use the stoichiometry of the balanced chemical equation to determine the number of moles of oxygen that react with the hydrogen chloride.
Finally, we can use the ideal gas law to determine the volume of oxygen that reacts with the hydrogen chloride.Let's begin by calculating the number of moles of hydrogen chloride:20.0 ml HCl x (1 L / 1000 ml) x (1 mol / 36.46 g) = 0.0005488 mol HCl
Now, we can use the stoichiometry of the balanced chemical equation to determine the number of moles of oxygen that react with the hydrogen chloride:4HCl + O2 → 2H2O + 2Cl20.0005488 mol HCl x (1 mol O2 / 4 mol HCl) = 0.0001372 mol O2
Finally, we can use the ideal gas law to determine the volume of oxygen that reacts with the hydrogen chloride: P = 1 atm (at standard temperature and pressure)R = 0.0821 L·atm/mol·KT = 273 K (at standard temperature and pressure)V = nRT / P = (0.0001372 mol) x (0.0821 L·atm/mol·K) x (273 K) / (1 atm) = 0.003 L = 3.0 ml
Therefore, the volume of oxygen gas that reacts with 20.0 ml of hydrogen chloride is 3.0 ml.
To learn more about chloride visit;
https://brainly.com/question/32108518
#SPJ11
Which property Is generally characteristic of an organic compound? (1) low melting point, (2) high melting point, (3) soluble In polar solvents, (4) Insoluble In nonpolar solvents.
Answers
General characteristic of an organic compound is low melting point. So, option (1) is correct.
What are organic compound?Organic compound are any of a large class of compounds in which one or more carbon atoms are covalently bonded to atoms of other elements, most commonly hydrogen, oxygen, or nitrogen. The only carbon-containing compounds not known to be organic include carbides, carbonates, and cyanides. Organic compounds are important because all living things contain carbon. They are the basic building blocks of many cycles that power the earth. For example, there is the carbon cycle, where carbon is exchanged between plants and animals during photosynthesis and cellular respiration.
Organic molecules have lower melting and boiling points because the attractive forces between molecules are weaker, so it does not require much energy to break them apart.
To know more about organic compounds, visit:
https://brainly.com/question/5994723
#SPJ1
1.54x10-1 L / 2.36x 10-45 solve this in scientific notation
Answers
Answer:
Fish is the best time to go
Explanation:
Chicken nuggets and a salad and some onion rings and a new charger
HELP LIKE HELP HURRY I HAVE 2 hours. To finish

Answers
Answer: hello, the answer would be rotates, revolves
hope this helps :)
Explanation:
Microscope Parts and Use Worksheet
Rack Stop
1
2
3
4
5
6
7
8
9
10
11
12
13
14
high
Microscope Part
power obiective into place

Answers
The Rack Stop is a small, movable metal stopper located at the bottom of the microscope body tube. Its purpose is to prevent the objective lenses from hitting the microscope slide or specimen, which could damage both the lenses and the sample.
Here is a list of microscope parts and their uses, including the Rack Stop:
1. Eyepiece or Ocular Lens: The lens at the top of the microscope that you look through to view the specimen.
2. Body Tube: The long, cylindrical part of the microscope that holds the eyepiece at the top and the objective lenses at the bottom
3. Arm: The curved part of the microscope that connects the body tube to the base.
4. Base: The flat, sturdy part of the microscope that supports the rest of the instrument.
5. Stage: The flat platform on which you place the specimen for viewing.
6. Stage Clips: Small metal clips that hold the microscope slide in place on the stage.
7. Coarse Focus Knob: A large knob that moves the body tube up and down to bring the specimen into rough focus.
8. Fine Focus Knob: A smaller knob that moves the body tube slightly to fine-tune the focus of the specimen.
9. Diaphragm: A rotating disc or lever that controls the amount of light entering the microscope and illuminating the specimen.
10. Light Source: The bulb or mirror that provides light for illuminating the specimen.
11. Objective Lenses: A set of lenses located at the bottom of the body tube that magnify the specimen.
12. Rack Stop: A small, movable metal stopper located at the bottom of the microscope body tube. Its purpose is to prevent the objective lenses from hitting the microscope slide or specimen.
13. Nosepiece: The rotating turret at the bottom of the body tube that holds the objective lenses.
14. High Power Objective: The objective lens with the highest magnification, typically 40x or higher. It is used for detailed examination of the specimen.
To use the microscope, first place the specimen on the stage and secure it with the stage clips. Turn on the light source and adjust the diaphragm to control the amount of light entering the microscope. Then, use the coarse focus knob to bring the specimen into rough focus. Once you have achieved this, use the fine focus knob to fine-tune the focus and bring the specimen into clear view. To change the magnification, rotate the nosepiece to select the desired objective lens. Finally, adjust the focus as needed and observe the specimen at the desired magnification.
For more questions on: metal
https://brainly.com/question/28183884
#SPJ11
what might happen if a low molecular weight carboxylic acid is exposed to a slightly acidic aqueous environment such as skin?
Answers
If a low molecular weight carboxylic acid, such as acetic acid, is exposed to a slightly acidic aqueous environment such as skin, it may undergo protonation and form its corresponding conjugate acid. This can lead to irritation or a burning sensation on the skin.
When a low molecular weight carboxylic acid is exposed to a slightly acidic aqueous environment like skin, the carboxylic acid may undergo the following processes:
1. Ionization: In the presence of water, carboxylic acid can ionize to form a carboxylate anion and a hydronium ion. This process is reversible, and the extent of ionization depends on the pKa of the carboxylic acid and the pH of the environment.
2. Partitioning: Due to its low molecular weight, carboxylic acid may be able to readily diffuse through the skin's aqueous layers. The partitioning of the carboxylic acid between the aqueous environment and the skin layers will depend on the compound's hydrophilicity or lipophilicity.
3. Interaction with skin proteins: Carboxylic acid may also interact with proteins in the skin, forming hydrogen bonds or other non-covalent interactions. These interactions can affect the overall skin properties, such as hydration, pH, and barrier function.
4. Possible irritation or sensitization: Depending on the specific carboxylic acid and its concentration, exposure to the skin may cause irritation or sensitization. This can result in redness, itching, or other signs of skin discomfort.
In summary, when a low molecular weight carboxylic acid is exposed to a slightly acidic aqueous environment such as skin, it may ionize, the partition between the aqueous environment and skin layers, interact with skin proteins and potentially cause irritation or sensitization.
Learn more about acids here: https://brainly.com/question/31050542
#SPJ11
Perform the followingmathematical operation, andreport the answer to thecorrect number of significantfigures.0.97584 = 5.68 = [ ?]=Enter
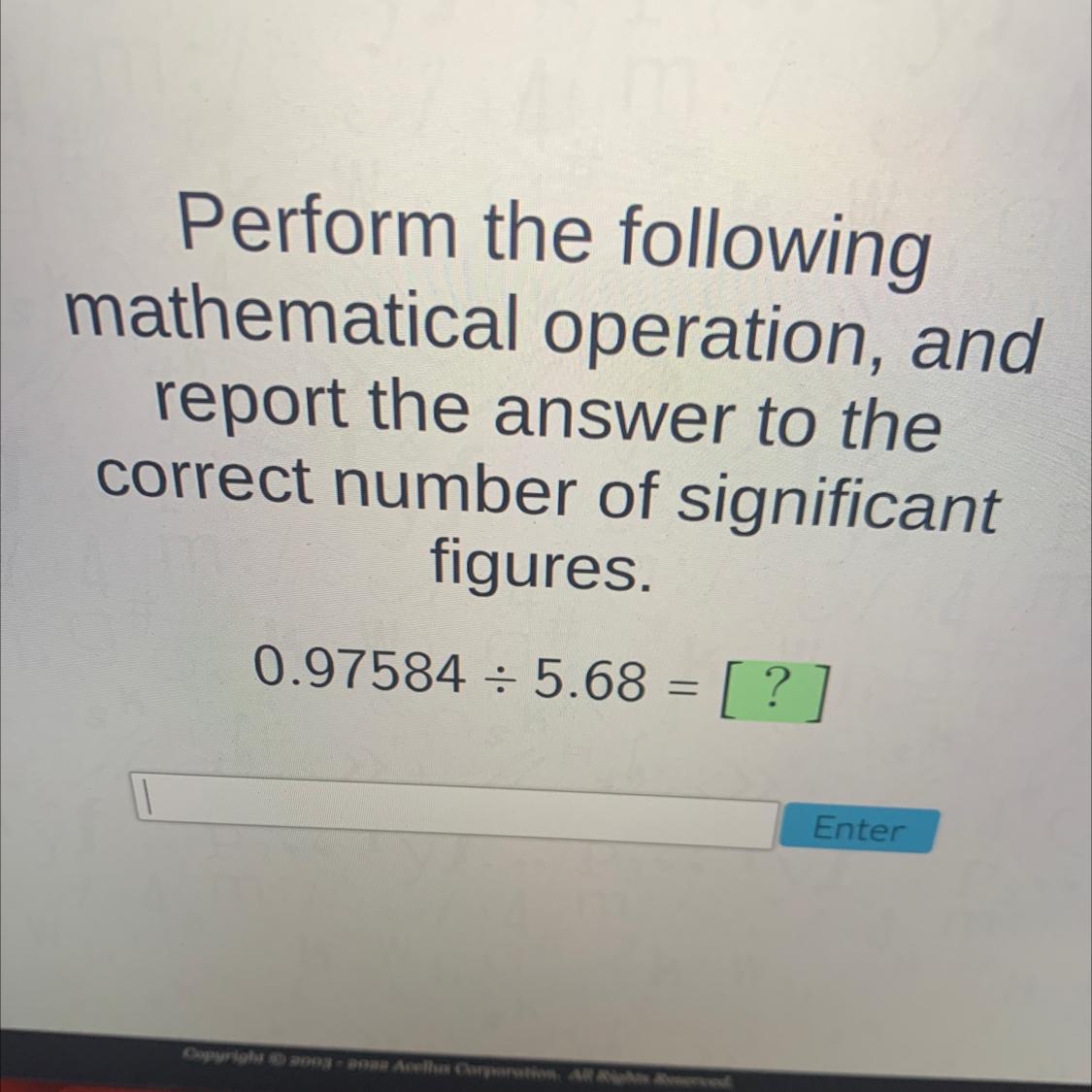
Answers
When we do the respective division operation we get the following result 0.171802.
When we do the respective division operation we get the following result 0.171802. But we must adjust the significant figures taking into account the number that has less significant figures, which is 5.68.
5.68 has three significant figures. So we will adjust the significant figures of the result to three.
0.97584 / 5.8 = 0.1718 = 0.172
what method will you use to separate the mixture of two solids which have different solubility in water
Answers
Answer:
A fractional crystallisation method is used for separating a mixture of two solids, if their solubilities in a particular solvent differ widely.
Explanation:
Describe what a mole is,
and what is meant by
a molar solution.
Answers
Answer:
A mole is an amount of a substance that contains 6.022✖️10^22 atoms of that substance
Or
Gram atomic masses,gram formula masses or gram formula masses are called one mole.
Molar solution or molarity is defined as the number of miles of so lutes dissolved per dm^3 of solution
Explanation:
I hope this will help you :)
If the number of moles in a balloon is doubled, but the temperature and pressure don't change, what happens to the volume?
a
It increases, but not in proportion with the moles
b
It is cut in half
c
It doubles
d
It does not chang
Answers
what is the pOH of a solution if the concentration of hydronium ionsis 8.9x10-9
Answers
pOH of a solution = 5.95
Further explanationGiven
The concentration of hydronium ions : 8.9 x 10⁻⁹
Required
pOH
Solution
pOH = - log[OH]
[H₃O⁺]= 8.9 x 10⁻⁹
pH = - log[H₃O⁺]
pH = 9 - log 8.9
pH = 8.05
pH + pOH = 14
pOH = 14-pH
pOH= 14-8.05
pOH = 5.95
Aqueous potassium chloride will react with which one of the following to form a precipitation reaction?
copper (II) nitrate
sodium chromate
ammonium phosphate
lead (II) chlorate
Answers
Answer:
lead (II) chlorate
Explanation:
precipitate means not soluble in water
potassium chlorate is not very soluble in water, it precipitates and may be collected by filtration
lead(II) chloride can be made as a white precipitate by adding a solution containing chloride ions to lead(II) nitrate solution
thoughtcocom chemguidecouk